Report 2010
Summary
Allocation of critical incidents in the adminsitration process
Reports are categorized in the various steps 1 to 5 in the process of blood product administration. Identification steps (1/5) are located at almost every step.
Where in the process the incident is located?
By far the most errors occur in the step of blood product administration. Secondary to that but still remarkable, are indication approval, handling & storage and the blood bank.
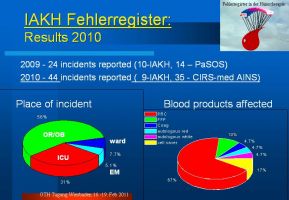
Where exactly in the hospital structure do incidents happen and which blood products are affected ?
More than half of incidents happen in the operation theater or the obstetric suite. In ICU, only a third of all reports are based in.
The handling and adminstration of allogeneic packed red cells is very prominent among all reported incidents (67% allogeneic, 5% autologous). Suprisingly high is the contribution of all reported incidents in cell salvage use (17%). Fresh frozen plasma did cause a report in 10% allogeneic, 5% autologous and coagulation concentrates/preparations in 5%. One error with the use of platelets have been reported (missing in the graph).
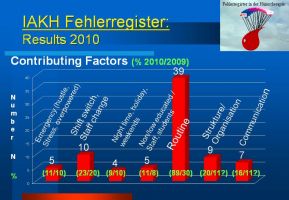
What are the contributing factors ?
Circumstances in which the incidents occurred in were analyzed. Almost 90% of all reported incidents happened during routine working conditions. In almost 23% a change of shifts was associated. Structural weaknesses or missing or failed communication amongst individuals or disciplines were contributing to the event in 9 and 7% of all reports.
What is the cause of the reported incident?
An identification error of the patient, the blood sample or the blood product was the underlaying reason in 37% of all incidents and stayed undetected in 15% of all reports. False indication for a blood product was detected form all reports in more than a tenth. The way blood sampling was performed was erroneous a fourth, the aminstration process in another fourth. The wrong or outdated blood product administration was undetected in 8%.
Recommendations given in reaction to the report
In general, all critical incidents shoud be discussed by the German "Transfusionskommission", a obligatory legal structure, that discusses the quality and organization of the local transfusion system. In most cases, the recommendation was to inform all contributors of an incident by an interdisciplinary education lecture about the erronous topic.
Remarkably, almost in 60% the error could have been avoided by the existence of an SOP, check list, protocol, or algorithm. A third of reported incidents could be avoided by an IT solution - the compatibility and communication of the blood banking software with the PDMS has the potential to avoid a quarter of all reported incidents!
Cases
(still in translation and entry process)
- 18-2010-t5e7-Insufficient blood units for major spine surgery case and known antibodies
- 19-2010-i9u8-Administration of platelets in a rapid infusion device
- 20-2010-N4E7-Near miss confusion of cell saver reservoir
- CM 5414-2010-Blood sample for type and screen in an unlabeled tube
- CM 5311-2010-Transfusion to the wrong recipient with the same blood group
- CM 4829-2010-Wrong hemoglobion content in blood gas sample
- CM 4918-2010-Jehovah`s Witness membership not detected
- CM 4789-2010-Confusion of blood unit in massive transfusion situation on ICU
